nstitutional Biosafety Committee
nstitutional Biosafety Committee
Purpose and Function
The Institutional Biosafety Committee (IBC) reviews and evaluates investigator-generated safety protocols for the proposed use of agents deemed hazardous by the NIH/CDC and UAMS Occupational Health & Safety (OH&S).
These reviews determine whether the IBC approves the safety protocols, with periodic reports to the Safety Coordinating Committee.
The IBC also makes recommendations to the Safety Coordinating Committee about biohazards that may exist or arise on the UAMS campus.
General Information
Organization and Responsibility
Research personnel with expertise in infectious agents, toxicology, recombinant DNA, animals, and/or human gene therapy make up most of the IBC membership. In accordance with NIH/CDC guidelines, members also include Biosafety Officers and two members who represent the interests of the community.
The UAMS Chancellor appoints committee members and a chair. The chair or chair’s designee signs off on behalf of the committee to approve biohazard safety protocols.
The committee meets at least quarterly or when convened.
Membership
IBC members include two community members, a non-doctoral member, and the UAMS Biosafety Officers. Other members have expertise in biosafety, animal science, virology, bacteriology, pathology, and (human) gene therapy.
Their membership and qualifications must comply with Sections IV-B-2-a and IV-B-2-b. Ad hoc members may be called upon for specific expertise.
Members terms are three years, which may be renewed. The UAMS Chancellor may limit or extend terms as needed.
The committee solicits nominations for membership. The Chancellor’s Office makes committee appointments, and the Chancellor appoints the committee chair.
Submission and Meeting Schedule
|
Specialist Review Due Date (2nd Friday of the Month) |
Committee Review IBC Agenda Set * (3rd Friday of the Month) |
IBC Meeting Date (1st Friday of Next Month) |
|---|---|---|
| 5/8/2026 | 5/15/2026 | 6/5/2026 |
| 6/12/2026 | 6/19/2026 | 7/3/2026 |
| 7/10/2026 | 7/17/2026 | 8/7/2026 |
| 8/14/2026 | 9/21/2026 | 9/4/2026 |
| 9/11/2026 | 9/18/2026 | 10/2/2026 |
| 10/9/2026 | 10/16/2026 | 11/6/2026 |
| 11/13/2026 | 11/20/2026 | 12/4/2026 |
| 12/11/2026 | 12/18/2026 | 1/1/2027 |
| 1/8/2027 | 1/15/2027 | 2/5/2027 |
| 2/12/2027 | 2/19/2027 | 3/5/2027 |
| 3/12/2027 | 3/19/2027 | 4/2/2027 |
| 4/9/2027 | 4/16/2027 | 5/7/2027 |
| * Note: Only complete protocols that meet the Muse Protocol Checklist requirements will be added to the IBC agenda. | ||
Proposals to use hazardous agents in research are submitted via Muse. Send any questions to the Institutional Biosafety Committee or the Biosafety Officers.
IBC meetings are held the first Friday of the month. Only Biosafety Officers place protocols on the meeting agenda, which is set two weeks before meetings to allow for IBC member review.
Regulations and Guidelines
- NIH Guidelines for Research Involving Recombinant or Synthetic Nucleic Acid Molecules , Department of Health & Humans Services, National Institutes of Health (PDF)
- Biosafety in Microbiological and Biomedical Laboratories (BMBL) 6th Edition, Centers for Disease Control and Prevention (CDC)
Hazardous Agents
The following items require review by the UAMS IBC:
- Infectious agents at biosafety level-2 (BL-2) or higher, including bacteria, viruses, mycotic agents, parasites, prions, and virus-shedding tumor cells.
- Highly toxic compounds.
- DNA recombination involving BL-2 or higher organisms or genes.
- Gene therapy of humans (whether or not the rDNA reagent is generated at UAMS). Under NIH Guidelines, Institutional Review Board approval of such human gene transfer clinical trials is contingent upon IBC approval
- Operating performance and SOPs of each newly-constructed or remodeled Biosafety Level 3 (BSL3) laboratory before it opens.
- Samples, tissues, or cells from humans or non-human primates.
IBC Monthly Meeting Minutes
- April 2026 IBC Minutes
- March 2026 IBC Minutes
- February 2026 IBC Minutes
- January 2026 IBC Minutes
- December 2025 IBC Minutes
- November 2025 IBC Minutes
- October 2025 IBC Minutes
- September 2025 IBC Minutes
- August 2025 IBC Minutes
- July 2025 IBC Minutes
- June 2025 IBC Minutes
Topics for Researchers
Muse IBC Protocol
The image below is an example protocol home page in Muse. Each Muse protocol follows this workflow.
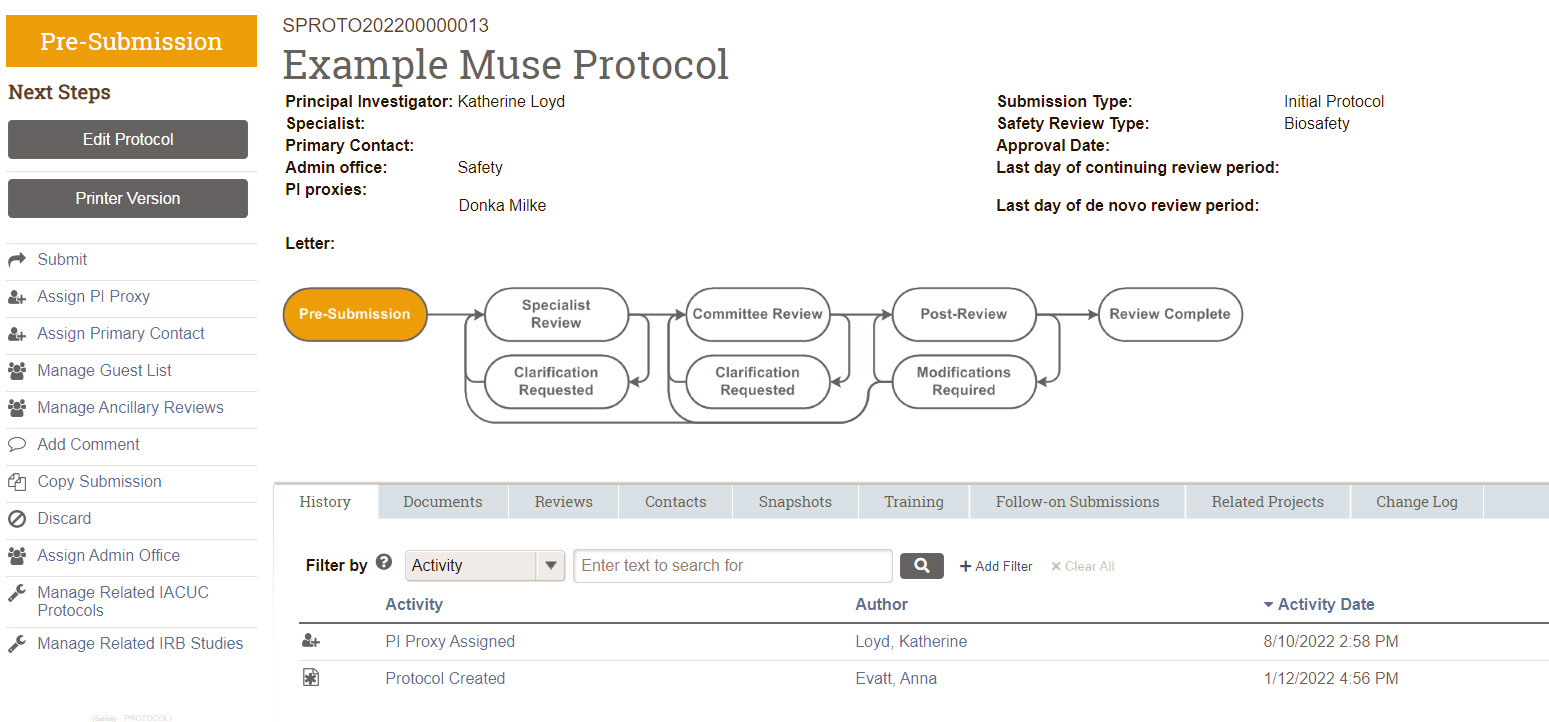
The following sections describe the steps that occur when you protocol is in each of the states in the headings.
Pre-Submission State (Draft)
-
To create your protocol in Muse, click Submit Your IBC Protocol
under the Biosafety Quick Links.
Note: Only the PI or PI Proxy may submit a protocol. - Click the Safety tab, then click Create Safety Submission.
- Follow prompts and complete all required fields until your protocol is complete according to the Muse Protocol Checklist rubric.
- Click Save while working on your protocol and use the Validate button to find incomplete sections.
- Once your protocol is complete, click Submit on the protocol home page and complete the Investigator’s Assurance form to officially begin the review process.
This action moves your protocol from the draft Pre-Submission state to Specialist Review state.
Note: Clicking Finish in the Supporting Documents section does not submit your protocol.
Specialist Review State (Review Begins)
-
In this state, your protocol must pass two pre-reviews before going to the IBC:
- Biosafety Review: A Biosafety Officer adds comments and summarizes review findings on the home page of your protocol.
- Final Specialist Review: This review is a final check of your protocol. If your protocol meets all the Muse Protocol Checklist requirements, the Biosafety Office will move the protocol onto the next IBC meeting agenda.
Note: You must address all biosafety review comments and complete any needed actions before the Final Specialist Review can occur.
Committee Review State (IBC Meeting)
-
In this state, the IBC Committee reviews your protocol and determines the next state:
- Approved moves the protocol to the Review Complete state, and an IBC approval letter is issued. (See more details below).
- Modifications Required moves the protocol to Post-Review and indicates the IBC requires protocol updates to gain approval. The required modifications display on the Muse home page for your protocol.
- Approval Withheld indicates the protocol does not meet safety and NIH guidelines and moves it to Post-Review state. Upon updating, the protocol will be returned to the Pre-Submission state to be resubmitted to the IBC.
- Deferred indicates the protocol does not include enough information for the committee to make an IBC ruling and it moves to Post-Review. Upon updating, the protocol will be returned to the Pre-Submission state to be resubmitted to the IBC.
Review Complete State
-
When your protocol reaches Review Complete, it is approved for a three-year De Novo
Period with required annual continuing reviews.
See your protocol home page in Muse for the:
- Annual Continuing Review (CR) due date. CRs must be completed annually to maintain IBC compliance.
- Three-Year De Novo Review due date.
Note: CRs must be completed annually to maintain IBC compliance.
Responsibilities of the Principal Investigator or Designee
Principal Investigators (PIs) are responsible for:
- Ensuring compliance with all institutional requirements, laws, and regulations for their research project.
- Ensuring safety and research compliance among all participants, staff, students, animal facility personnel, and others who may encounter their laboratory hazards through appropriate hazard notification and training
- Detailing procedures for the safe handling, storage, and disposal of hazardous agents they propose using according to IBC and OH&S guidelines. PIs may also be required to provide other information on request.
- Developing a risk assessment for each proposed project to determine containment requirements, set the required biological safety level of each project, and establish conditions they must comply with to conduct projects.
- Setting policy for the acquisition and safe handling, transfer, and storage of biohazardous agents, materials, and recombinant DNA constructs.
NOTE: PIs can assign a designee, such as a lab manager, using the PI Proxy option in Muse. However, this designation must be described in the Muse protocol, and the PI bears ultimate responsibility for all aspects of the research project.
Forms, Manuals, and Policies
Use the Muse Protocol Checklist below as a rubric for your protocol and the following information to assist you in your protocol management.
- Muse Protocol Checklist (PDF)
- Muse Additions (Link to Microsoft Sway)
- Continuing Review Instructions (PDF)
- Amendment Instructions (PDF)
- Committee Amendments: require full committee review due to scientific, hazard, class, or other experimental changes.
- Administrative Amendments: do not require full committee review. This is an expedited amendment for personnel, locations, or other administrative changes.
- Biosafety Required Trainings (PDF)
- Arkansas Children's Training Documentation
- Muse Protocol Example (PDF)
Note: Only approved protocols can be amended. Only one amendment can be submitted at a time. There are two amendment types:
Note: More information is available on the OH&S Biological Safety Division web page. Please contact the Biosafety Officers if you need assistance inputting your protocol.
Contacts
- IBC General Email: Institutional Biosafety Committee
- IBC Chairman: Jia Liu, Ph.D., Associate Professor, Department of Microbiology and Immunology
- Biosafety Officer: Lindsey Clark, MPH, MLS(ASCP)CM
- Biosafety Officer: J.C. Douglas, M.Sc., CCRP (SOCRA)
Biosafety Quick Links
Biosafety Quick Links
4301 W. Markham Street, Slot #617, Little Rock, AR 72205
Phone: (501) 686-5536
Campus Operations Call Center • Phone: (501) 526-0000 • 24 hours a day / 7 days a week